
In recent years, public attention has increasingly turned toward the behind-the-scenes processes that shape modern medicine. For many people, the journey of a drug—from discovery to pharmacy shelf—remains largely invisible. Yet occasionally, reports and images emerge that bring this hidden world into sharper focus, prompting questions about how treatments are developed and what role animals play in that process.
The conversation is not simple. It sits at the intersection of science, ethics, and public responsibility. While some view animal testing as a necessary step in ensuring safety, others question whether evolving technologies can offer more humane and equally effective alternatives.
Understanding this issue requires looking beyond headlines and exploring both perspectives with clarity and balance.

Why Animal Testing Exists
Before a new medicine reaches human clinical trials, it must undergo extensive safety evaluations. Regulatory agencies such as the U.S. Food and Drug Administration and the European Medicines Agency require data on how a substance behaves in a living system.
These studies aim to answer key scientific questions:
How is the substance absorbed and distributed in the body
How is it metabolized and eliminated
What effects might it have on organs and biological systems
What dosage levels are considered safe
Animal models have traditionally been used because they allow researchers to observe complex biological interactions that cannot yet be fully replicated in simpler systems.
According to organizations like Understanding Animal Research, this approach has contributed to the development of many widely used treatments, including therapies for chronic illnesses and preventive medicines.
Ethical Concerns and Public Debate
At the same time, the use of animals in research raises ethical concerns that continue to shape public debate.
Critics argue that animals can experience distress and discomfort during certain procedures, and they question whether such practices are justified—especially when alternatives may exist. Advocacy groups such as the Humane Society International emphasize the need to reduce reliance on animal testing and promote more humane approaches.
In response, many countries have implemented strict regulations designed to minimize harm. In the United Kingdom, for example, research is governed by laws that require justification for animal use, oversight by ethics committees, and adherence to welfare standards.
A widely accepted framework guiding these efforts is the principle of the “3Rs”:
Replacement of animals with alternative methods where possible
Reduction in the number of animals used
Refinement of procedures to minimize discomfort
These principles aim to balance scientific progress with ethical responsibility.
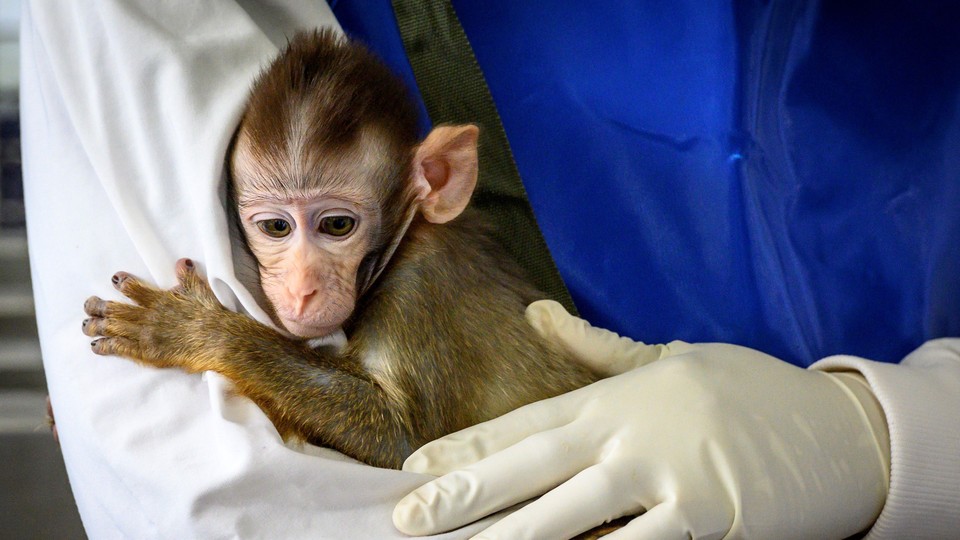
Scientific Limitations of Animal Models
Beyond ethics, there is also an ongoing scientific discussion about the limitations of animal testing.
Research highlighted by the National Institutes of Health suggests that biological differences between species can affect how accurately results translate to humans. Some treatments that appear promising in animal studies do not always produce the same outcomes in human trials.
This has led scientists to explore whether alternative methods might offer more precise predictions of human responses.
However, many experts caution that while alternatives are advancing, they do not yet fully replicate the complexity of a complete living organism. This is why animal testing remains part of the current system, even as new approaches are developed.
Emerging Alternatives: A New Era of Research
One of the most promising developments in recent years is the rise of human-centered research technologies.
The U.S. Food and Drug Administration has begun encouraging the use of innovative methods that may reduce reliance on animal models. These include:
Organ-on-a-chip systems that simulate human organ functions
Lab-grown tissues known as organoids
Advanced computer modeling and artificial intelligence
These technologies aim to replicate human biology more accurately while reducing ethical concerns. Scientists believe that combining these tools with traditional methods could lead to safer and more efficient drug development.
While still evolving, these approaches represent a significant shift in how research may be conducted in the future.
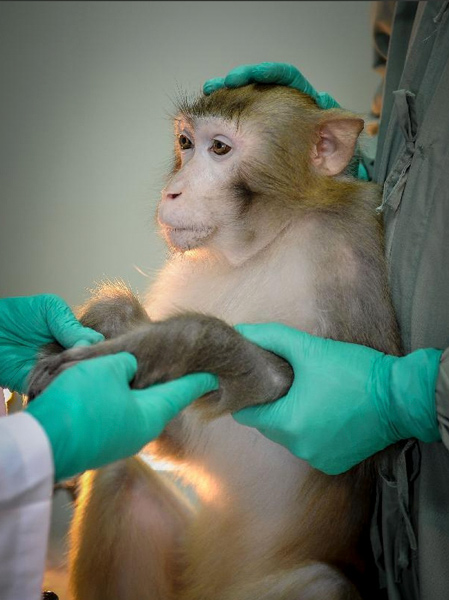
The Role of Regulation and Oversight
Regulatory frameworks play a crucial role in shaping how animal testing is conducted.
In the UK, research facilities operate under strict licensing and inspection systems. Similar oversight exists in other regions, ensuring that studies meet established ethical and scientific standards.
These systems are designed to ensure that animal use is justified, carefully monitored, and limited to situations where no viable alternative exists.
Transparency has also become an important focus. Public awareness and open discussion help build trust and encourage continued improvement in research practices.
Public Perception and Media Influence
Media coverage often plays a significant role in shaping how people view animal testing.
Images or reports can evoke strong emotional responses, especially when they highlight difficult aspects of research. At the same time, advocacy groups and scientific organizations may present different interpretations of the same issue.
This contrast underscores the importance of evaluating information critically and seeking balanced perspectives. Understanding both the scientific purpose and the ethical implications allows for more informed discussions.
A Complex Balance Between Progress and Responsibility
The debate over animal testing is not easily resolved. It involves weighing the potential benefits of medical advancements against concerns about animal welfare.
On one side, researchers emphasize the importance of ensuring that new treatments are safe and effective before they are used by humans. On the other, advocates push for faster adoption of alternatives that reduce or eliminate the need for animal involvement.
Both perspectives share a common goal: improving health outcomes while maintaining ethical standards.
The Future of Medical Research
Looking ahead, the direction of medical research appears to be moving toward greater integration of alternative methods.
Advances in technology are expanding what is possible, offering new ways to study human biology without relying solely on animal models. At the same time, regulatory agencies are adapting guidelines to support these innovations.
The transition will likely take time. Scientific validation, infrastructure development, and global coordination are all required to implement new approaches effectively.
However, the momentum is clear. The future of research is likely to involve a combination of methods, each contributing to a more comprehensive understanding of health and disease.

A Reflection on Curiosity and Responsibility
At its core, this topic reflects a broader human challenge: how to pursue knowledge responsibly.
Curiosity drives scientific discovery. It leads to breakthroughs that improve lives and expand understanding. But curiosity must also be guided by ethical consideration, ensuring that progress does not come at an unnecessary cost.
The discussion around animal testing invites us to think critically about how decisions are made, how technologies evolve, and how values shape scientific practice.
In the end, it is not just about choosing one path over another. It is about continuing to ask questions, seek better solutions, and move forward with both intelligence and compassion.
Sources
U.S. Food and Drug Administration (FDA)
European Medicines Agency (EMA)
National Institutes of Health (NIH)
Understanding Animal Research
Humane Society International